|
19/03/2022 09:45:41 |
|
Normal
|
|
Acknowledged
|
|
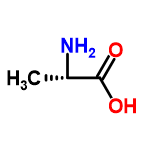
|
|
|
|
24/03/2022 10:39:21 |
|
The L/D notation can be confusing – L amino acids are not necessarily levorotatory. See this explanation (from https://chem.libretexts.org/Bookshelves/Biological_Chemistry/Supplemental_Modules_(Biological_Chemistry)/Proteins/Amino_Acids/Properties_of_Amino_Acids/Stereochemistry_of_Amino_Acids):
"All common amino acids are the L-enantiomer (i.e. their Cα chiral center is the L-enantiomer), based on the structural comparison with L-glyceraldehyde. However, not all L-amino acids are Levorotatory, some are actually Dextrorotatory with regard to their optical activity. To (attempt) to avoid confusion, the optical activities are given as (+) for dextrorotatory, and (-) for levorotatory
L(+)-alanine (this is the L-enantiomer and it is dextrorotatory)
L(-)-serine (this is the L-enantiomer and it is levorotatory)"
|